Topic outline
-
-
Forum
-
Course Outline
· Course Details
Course Title
Chemical Applications of Group Theory
Course Number
423522
Academic Year
2021-2022
Term
Summer Course
Prerequisite(s)
Departmental Approval
Course Type: Compulsory / Elective… etc.
Compulsory
Credit Hours
3
· Instructor Information
Instructor Name
Hikmat S. Hilal
Office
14F1310
Email Address
Mobile& Whatsapp: 00972599273460
· Class Details
Days
Sunday + Tuesday
Time
2-5
Class Room
· Course Description and Objectives
The course starts with terminology and definitions of molecular symmetry operations, matrix representation, point groups, reducible and irreducible representations, character tables and reduction equation. Applications of group theory to different aspects of chemistry, specially bonding, structure and spectroscopy, are rigorously included.
The course helps the student understand basics of group theory and its applications to different aspects of chemistry. Bonding theory, metal clusters, vibrational spectroscopy, absorption spectroscopy and organic reactions are rigorously involved.
· Intended Learning Outcome (ILO’s)
Upon completing this course, the students are expected to able to:
1) Implement details of molecular symmetry including symmetry elements, operations and symmetry point groups to different chemical compound structures
2) Use necessary mathematical basic information in group theoretical applications, including matrices, reduction formula, reducible and irreducible representations
3) Apply group theory in valence bond theory treatment of structure and bonding
4) Apply group theory in molecular orbital theory treatment of bonding and structure
5) Apply group theory in studying vibrational spectra of different inorganic compounds.
6) Use Group Theory in metal-metal cluster compounds (metal carbonyls and quadrupole bonds)
7) Apply group theory to predict concerted organic reactions.
· Textbook(s) and References
1) Hikmat S. Hilal & Abdel-Hafez Sayda, A Short Course on Group Theory and Chemical Applications, Novascience Publ., 2011.
2) A. Vincent, Molecular Symmetry and Group Theory, 2nd ed, (2000), J. Wiley.
3) F.A. Cotton, Chemical Applications of Group Theory, 3rd ed, (1990), J. Wiley; ANU Library Code 541.22015122 COT
4) J. Barrett, Introduction to Atomic Molecular Structure, J. Wiley
5) I. Fleming, Frontier Orbitals and Organic Chemical Reactions, J. Wiley
6) G.B. Gill & M. R. Willis, Pericyclic Reactions, Chapman
7) B. Douglas, D. H. McDaniel, J. J. Alexander, Models and Concepts of Inorganic Chemistry, 3rd ed., (1994), J. Wiley & Sons,
8) R. L. Carter, Molecular Symmetry and Group Theory, (1997), J. Wiley & Sons, ISBN-10: 0471149551.
9) FA Cotton and G Wilkinson, Advanced Inorganic Chemistry, J. Wiley.
10) F.A. Cotton, Chemical Applications of Group Theory, 3rd ed, (1990), J. Wiley; ANU Library Code 541.22015122 COT
11) Library search and primary sources.
· Topics Covered / Weekly Lecture Schedule
Week
Topics
1
Symmetry operations
2
Symmetry point groups
3
4
Representations, Character Tables, Reduction Formulas, and classes
Application in Valence Bond theory (VBT) & hybrid orbitals, and Molecular Orbital Theory
1st Test
5
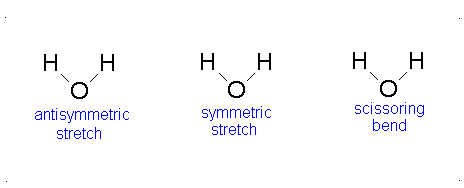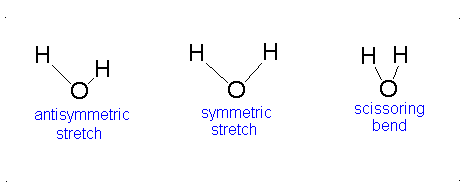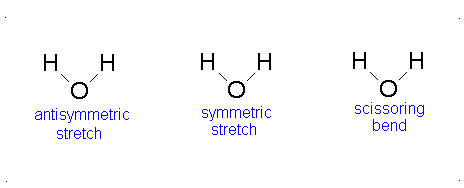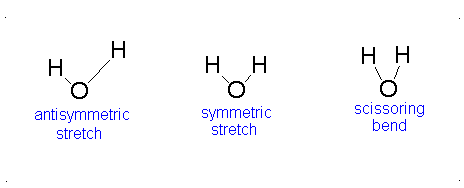
Applications in Vibrational Spectra (IR and Raman Spectra)
6
Applications in Molecular Orbital Theory: Diatomics, triatomics (Walsh Diagrams), and complexes,
7
8
9
10
Applications in organic cyclic systems, Huckel’s aromaticity, conjugated systems
2nd Test
11
12
13
Metal atom clusters (metal carbonyls and noncarbonyls)
14
Applications in concerted organic reactions
15
16
Final Exam
· Assessment Measures and Methods of Evaluation
Midterm 30%
Activities 20%
Final Exam 50%
· Important Dates
HSH Updated: June 2nd, 2022
-
Topic One :Students Independent Reading
-
-
-
Topic Four :Representations, character Tables, Reduction Formulas, and classes
-
Topic Five:Application in Valence Bond theory (VBT) & hybrid orbitals, and Bonding TheoryFile
-
Topic Six:Application to Vibrational SpectroscopyFile
-
-
Topic Eight:Triatomic Systems.
-
Topic Nine :AB3 Molecular SystemsFile
-
Topic Ten:Huckel's AromaticityFile
-
Topic Eleven :Metal atomic clusters (Non-carbonyls)File
-
Topic Twelve : Group Theory in Organic Pericyclic Reactions
-
-
https://drive.google.com/drive/folders/1wXKKadQgzmJFGsSdWJyoiFWdCLngXkdi?usp=sharing
-
https://drive.google.com/drive/folders/1TIqgZoGXU6TLj-hgCXsXMQoDvPvSdDP2?usp=sharing
-
-
-
Assignment
1) Consider the systems: [NiCl4]2- and [Cr(H2O)6]2+ . For each system,
- Show a molecular structure, and write down symmetry point group
- Use Group theory to find suitable hybrid orbitals of the central atom Sigma bonds
- Us Group theory to find the Sigma bond Molecular Orbital Energy Level Diagram for the [NiCl4]2- ion
- Describe how Group theory and Vibrational spectra can help confirm the structure for the [NiCl4]2- ion (Td or D4h)
-